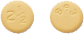ELIQUIS may cause serious side effects, including increased risk of blood clots if you stop taking ELIQUIS. ELIQUIS lowers your chance of having a stroke by helping to prevent clots from forming. Do not stop taking ELIQUIS without talking to the healthcare provider who prescribed it for you. Stopping ELIQUIS increases your risk of having a stroke.
ELIQUIS may need to be stopped prior to surgery or a medical or dental procedure. Your healthcare provider will tell you when you should stop taking ELIQUIS and when you may start taking it again. If you have to stop taking ELIQUIS, your healthcare provider may prescribe another medicine to help prevent a blood clot from forming.
This is a summary of important safety information that you need to know about ELIQUIS. Keep this document in a safe place, so you can refer to it before and during your treatment.
Look out for the following icons as you read:

Talk to your
healthcare team

Call a healthcare
provider right away

Helpful information
to remember
What is the most important information I should know about ELIQUIS?
ELIQUIS may cause serious side effects, including:

Increased risk of blood clots if you stop taking ELIQUIS
ELIQUIS lowers your chance of having a stroke by helping to prevent clots from forming. If you stop taking ELIQUIS, you may have an increased risk of a clot forming in your blood.

![]() Do not stop taking ELIQUIS without talking to the healthcare provider who prescribed it to you.
Do not stop taking ELIQUIS without talking to the healthcare provider who prescribed it to you.
Stopping ELIQUIS increases your risk of having a stroke.

Talk to your healthcare team before any medical procedures. ELIQUIS may need to be stopped before surgery or a medical or dental procedure. Your healthcare provider will tell you when you should stop taking ELIQUIS and when you may start taking it again. If you have to stop taking ELIQUIS, your healthcare provider may prescribe another medicine to help prevent a blood clot from forming.

Bleeding
ELIQUIS can cause bleeding, which can be serious, and may lead to death. This is because ELIQUIS is a blood thinner medicine that reduces blood clotting. While taking ELIQUIS, you may bruise more easily and it may take longer than usual for any bleeding to stop.
You may have a higher risk of bleeding if you take ELIQUIS and take other medicines that increase your risk of bleeding, such as:

Tell your healthcare provider if you take any of these medicines.

Call your healthcare provider or get medical help right away if you or your child have any of these signs or symptoms of bleeding when taking ELIQUIS:
Bleeding that is severe or you cannot control
Red, pink, or brown urine
Red or black stools (looks like tar)
Coughing up or vomiting blood or blood clots
Vomit that looks like coffee grounds
Unexpected pain, swelling, or joint pain
Headaches
Feeling dizzy or weak

Spinal or epidural blood clots (hematoma)
People who take a blood thinner medicine like ELIQUIS, and have medicine injected into their spinal or epidural area, or have a spinal puncture, have a risk of forming a blood clot that can cause long-term or permanent loss of the ability to move (paralysis).
Your risk of developing a spinal or epidural blood clot is higher if:
If you take ELIQUIS and receive spinal anesthesia or have a spinal puncture, your healthcare provider should watch you closely for symptoms of spinal or epidural blood clots or bleeding.

Tell your healthcare provider right away if you have any of these signs or symptoms when taking ELIQUIS:

Talk to your healthcare provider about any side effect that may be bothering you or that does not go away. These are not all of the possible side effects of ELIQUIS. Call your healthcare provider for medical advice about side effects.
You are encouraged to report any side effects to the FDA at 1-800-FDA-1088.
Who should not take ELIQUIS?

ELIQUIS is not for patients who:
What should I discuss with my healthcare team before starting ELIQUIS?

Talk to your healthcare team if you or your child:

Tell your healthcare provider right away if you become pregnant during treatment with ELIQUIS. Taking ELIQUIS during pregnancy may increase the risk of bleeding in you or your unborn baby.
Females who are able to become pregnant: talk with your healthcare provider about pregnancy planning, and your risk of severe uterine bleeding if you are treated with ELIQUIS.
Tell your healthcare provider if you are breastfeeding or plan to breastfeed. It is not known if ELIQUIS passes into your breast milk. Do not breastfeed during treatment with ELIQUIS. Talk to your healthcare provider about the best way to feed your baby during treatment with ELIQUIS.

Tell your healthcare team about all the medicines you or your child take, including any:

During treatment, make sure you talk to your healthcare team before you begin to take any new medication.
What are the possible serious side effects of ELIQUIS?
The most common side effect of ELIQUIS in adults was bleeding.
The most common side effects of ELIQUIS in children include: headache, vomiting, heavy menstrual bleeding.
How should I take ELIQUIS?

Take ELIQUIS exactly as prescribed by your healthcare provider.

Twice daily

Do not change your dose or stop taking ELIQUIS unless your healthcare provider tells you to.
What if I miss a dose of ELIQUIS?

If you or your child misses a dose of ELIQUIS, take it as soon as you remember on the same day. Do not take two doses of ELIQUIS at the same time to make up for a missed dose. Return to your usual dosing schedule the next day.
For children who take ELIQUIS:
See the detailed Instructions for Use that comes with ELIQUIS SPRINKLE for oral suspension or ELIQUIS tablets for oral suspension for information on how to prepare and give a dose of ELIQUIS to children who weigh less than 77 pounds (35 kg).
Always give ELIQUIS exactly as your child’s healthcare provider or pharmacist has told you.
The dose of ELIQUIS is based on your child’s body weight. Your child’s healthcare provider may need to change your child’s dose as their weight changes. Do not change your child’s dose without talking with the healthcare provider.
If your child vomits or spits up within 30 minutes after taking ELIQUIS, repeat the dose. If your child vomits or spits up more than 30 minutes after taking ELIQUIS, do not repeat the dose. Continue to give the next ELIQUIS dose at the next scheduled time.
Contact the healthcare provider if your child repeatedly vomits or spits up after taking ELIQUIS.
When should I refill my prescription?

Do not stop taking ELIQUIS without first talking with your healthcare provider. Do not run out of ELIQUIS. Refill your prescription before you run out. When leaving the hospital following hip or knee replacement, be sure that you will have ELIQUIS available to avoid missing any doses.
ELIQUIS is available in two different tablets. Your healthcare provider will determine what dose is right for you.
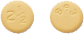
2.5 mg tablet

5 mg tablet
On one side of each film-coated tablet, there is a number indicating the dosage strength of the tablet (in milligrams). On the other side, there is a medication identification number.
ELIQUIS is also available as ELIQUIS SPRINKLE, a capsule containing a powder for oral suspension, or as a film-coated tablet for oral suspension for pediatric patients.

0.15 mg capsule

0.5 mg tablet
The capsule is printed with a medication identification number.
What is ELIQUIS?
ELIQUIS is a prescription medicine used in adults to:

Reduce the risk of stroke and blood clots in people who have atrial fibrillation (AFib), a type of irregular heartbeat, not caused by a heart valve problem.

Treat blood clots in the veins of the legs (deep vein thrombosis – DVT) or lungs (pulmonary embolism – PE), and reduce the risk of them occurring again after receiving treatment for blood clots.

Help prevent a blood clot in the legs (DVT) and lungs (PE) of people who have just had hip or knee replacement surgery.
ELIQUIS is a prescription medicine used in children from birth and older to:

Treat blood clots in the veins of the legs and lungs (venous thromboembolism) after at least 5 days of initial coagulant treatment, and reduce the risk of them occurring again.
ELIQUIS was not studied and is not recommended in children who weigh less than 5.7 pounds (2.6 kg).
The product information provided in this site is intended only for residents of the United States. The products discussed herein may have different product labeling in different countries.
The health information contained herein is provided for educational purposes only and is not intended to replace discussions with a healthcare provider. All decisions regarding patient care must be made with a healthcare provider, considering the unique characteristics of the patient.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.